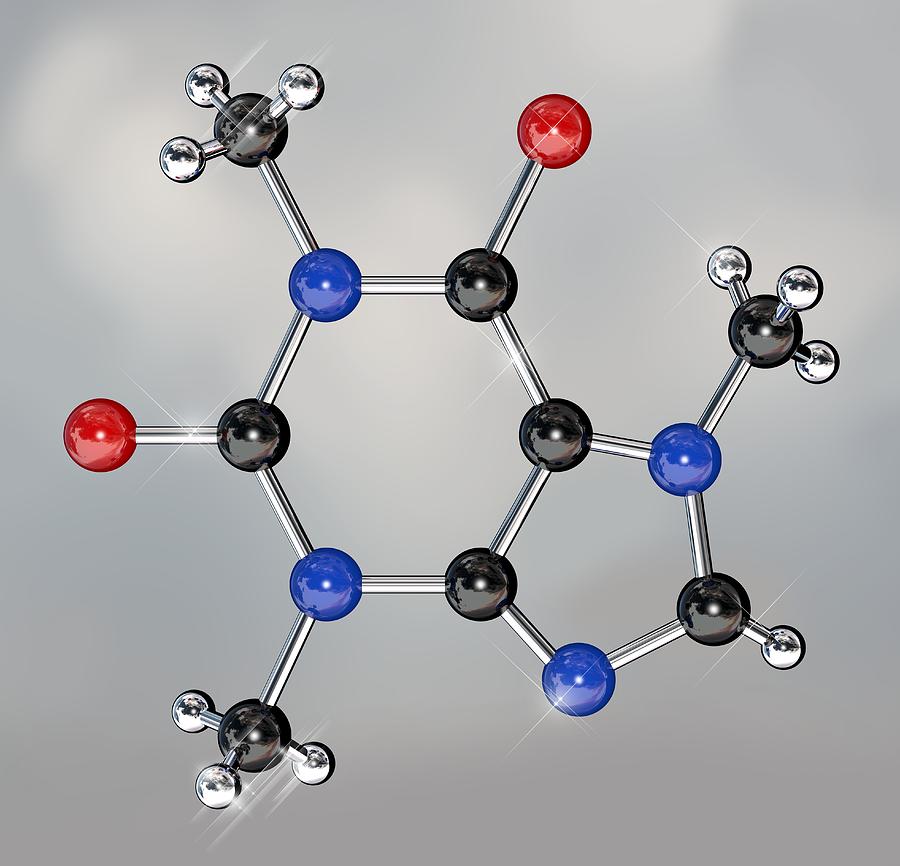
Although supercritical carbon dioxide is non-polar, and should only be able to dissolve non-polar substances, certain co-solvents, like water, can be added so that supercritical carbon dioxide can actually dissolve polar molecules like caffeine. It behaves like gas, and permeates a porous substance, while also exhibiting liquid properties to dissolve substances. This method is repeated until the coffee beans are 99.9% decaffeinated, and the flavor is left intact.Ĭarbon dioxide supercritical fluid (temperature above 31.1 ☌ and pressure above 73 atm) exhibits both liquid and gas-like behavior. Since the only difference between the mixture and the fresh coffee beans is the caffeine concentration, caffeine molecules will diffuse out of the beans into the mixture of coffee solids, leaving the coffee beans caffeine free. Concentration gradients take advantage of the law of simple diffusion- the movement of molecules from an area of high solute concentration to an area of low solute concentration in order to 'even out' the uneven distribution of molucules. The mixture creates a concentration gradient when added to a fresh batch of coffee beans. After filtration, the mixture that is left is water saturated with coffee flavor molecules – referred as “coffee solids” by the manufacturers. The activated carbon filter has holes large enough to allow water and coffee solids (smaller molecules) to pass through, but not caffeine (relatively larger molecule). The carbon filter is made out of activated carbon, carbon that has been made porous through the process of carbonization (reacting carbon in anaerobic conditions until the gaps between carbon atoms are large enough to allow molecules to pass through). The beans are then discarded, and the solution of water, caffeine, and coffee solids is passed through a carbon filter. First, unroasted (green) coffee beans are soaked in water until caffeine is dissolved in water. The Swiss Water Processing method removes caffeine without using any chemicals, but instead applies the law of simple diffusion. Therefore, this compound should be the best choice for this company. In addition, Dimethyl Sulfoxide dissolves caffeine better than other substances, which means its consumption will be lower. 
Dimethyl Sulfoxide was chosen as the best solvent because it meets the requirements. A particular solvent for decaffeinated beverages should be based on company preferences, namely environmental safety, and physical and chemical properties. In conclusion, it is worth emphasizing that developing a beverage requires a comprehensive approach. Consequently, Dimethyl Sulfoxide has all the necessary characteristics: it is environmentally friendly, polar, and an excellent dissolving agent. 
The solubility of caffeine in Dimethyl Sulfoxide is higher than in all other variants. In addition, the fourth type of solvent, Dimethyl Sulfoxide, which is moderately polar and non-toxic, has been proposed (ACS, 2021). 
The figure below shows that Methylene Chloride has the best solubility, Ethyl Acetate comes second, and caffeine dissolves the worst in water. Water and Ethyl Acetate have the molecule’s polarity, so their use is preferable.įinally, the last criterion is the solubility of caffeine in different media. Only Methylene Chloride is not a polar molecule, so it is not the best choice for making a drink. This will create the basis for faster and better solubility of the alkaloid in Decaf. Since the caffeine molecule has a slight polarity, the solvent should also be polar to increase the force of attraction between them. In other words, from an environmental perspective, water is the best choice.Īny dissolution process is primarily the physical stirring of caffeine in a solvent medium. Finally, Ethyl Acetate is a less toxic substance but can cause dizziness, drowsiness, and irritability with prolonged use (ACS, 2019). The CDC (2018) reports Methylene Chloride to be a carcinogen and an irritant, but trace amounts of the remaining solvent after drying are significantly lower than the FDA-approved standard. Pure water has no toxic properties whatsoever. Consequently, the choice of solvent must be based on the toxic properties of each of the substances. This is an excellent marketing and socially conscious commercial practice to attract consumers. One of the main arguments of the hypothetical company is the desire to make the final beverage environmentally friendly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
